
Proton number determines the number of electrons in an atom, which influences the number of electrons that can be gained or lost during a chemical reaction. On the contrary, weak attraction causes the nucleus to let go of its outermost electrons. If the attraction is felt strongly by the electrons, the nucleus can pull even more electrons in. The rms-radius R of the proton charge distribution is a fundamental quantity needed for precision physics. 
it is electrons that just move and decides positive and negative charge on body'. we do not use proton excess or deficiency terminologies because protons are way more heavier than electrons and do not move when we charge a body either negative or positive. The number of protons influences the positive charge of the nucleus, causing it to attract an equal number of negatively charged electrons. 'deficiency of electrons results in positive charge on body, while excess of electrons means body have negative charge. An elementary charge is denoted by the letter e and was originated in 1874 by George Stoney. However, we can see the protons of an atom as the puppeteer pulling the strings behind the scene. Protons have a charge of 1 elementary charge. It is the electrons that get passed around or shared between atoms. Field PHY 2049 Chapter 22 chp222. Indeed, protons remain unchanged in a chemical reaction. Electric Charge : electron charge -e e 1.6x10-19C proton charge e C Coulomb Electric charge is a conserved quantity (net electric charge is never created or destroyed ) q1q2 r R. If protons are hidden in the nucleus and bound tightly by the strong force, how do they influence a chemical reaction and hence shape the chemical identity of an atom? Protons are the invisible hand behind a chemical reaction This explains why any element with 2 or more protons necessarily has neutrons in their nucleus.Īll the protons of an atom are concentrated in the nucleus. Lo and behold, this stronger force is called the strong force, which is mediated by the neutrons. This suggests that is another extraordinary force holding the protons together, stronger than the electrostatic repulsion. Perhaps imagine squeezing the entire school into a classroom? The packing is way more intense than how students are squeezed into the school hall for assemblies. They realised that the protons of an atom are squeezed within the narrow confines of the nucleus. Yet, the earnest work of Ernest Rutherford and his colleagues in the 1900s blew everyone’s mind. Since like charges repel, early scientists expected protons to be spread out in an atom. $0.8409 \pm 0.0004 fm$ determined from the Lamb shift in muonic hydrogen.Even though iron-56 has 26 protons, they are all held very tightly together in the nucleus by the strong forceĪ proton has a relative charge of 1 . This value disagrees with the more precise value precision spectroscopy of atomic hydrogen, and. There are two completely different sources of experimental information on the proton charge rms radius r p. In the high-$q$ data, the model dependence of the extrapolation is reduced andĭifferent parameterizations of the pre-2010 data yield a consistent value for The rms-radius of the proton charge distribution is a fundamental quantity needed for precision physics. The charge mean-square radius of the proton is given by (2) r E p 2 0 r 2 c h ( r) 4 r 2 d r and (3) r E p 2 6 d G E p ( t) d t t 0. Knowledge about this large-$r$ density together with the information contained Parameterized $G(q)$ needed for the extrapolation. This quantity is expressed in femtometres (1 fm is 10 15 metres). 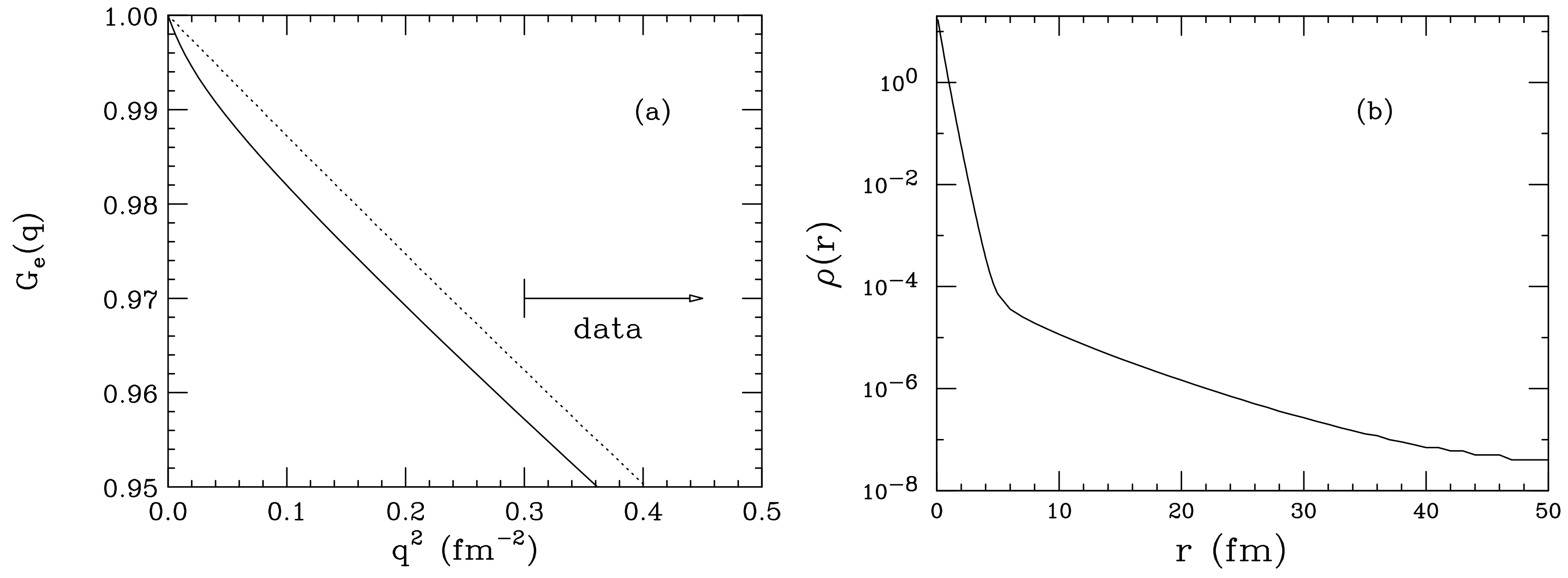
In addition to the electron, all freely existing charged subatomic particles thus far discovered have an electric charge equal to this value or some whole-number multiple of it. For example, electrons have negative charge and protons have positive charge, but neutrons have zero charge. electron charge, (symbol e ), fundamental physical constant expressing the naturally occurring unit of electric charge, equal to 1.602176634 × 10 19 coulomb. The problem lies in the model dependence of the A key property of the proton is its charge radius the spatial extent of its charge distribution. Britannica Quiz Physics and Natural Law Many fundamental, or subatomic, particles of matter have the property of electric charge. In order to identify the sources of the discrepancies, and explore alternative Weĭiscuss the approaches used to analyze the e-p data, partly redo these analyses $G_e(q^2)$ extrapolated to momentum transfer $q^2$=0, shows a large scatter. to carry out a meticulous study of systematic uncertainties and to extract a precise value for the proton radius: 0.833 ± 0.010 femtometres (1 fm is 10 15 metres). This radius, traditionally determinedįrom elastic electron-proton scattering via the slope of the Sachs form factor Download a PDF of the paper titled Proton charge radius from electron scattering, by Ingo Sick Download PDF Abstract: The rms-radius $R$ of the proton charge distribution is a fundamental
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
